MITOCHONDRIAL EXOMARKERS AND LC-MS/MS PROBES
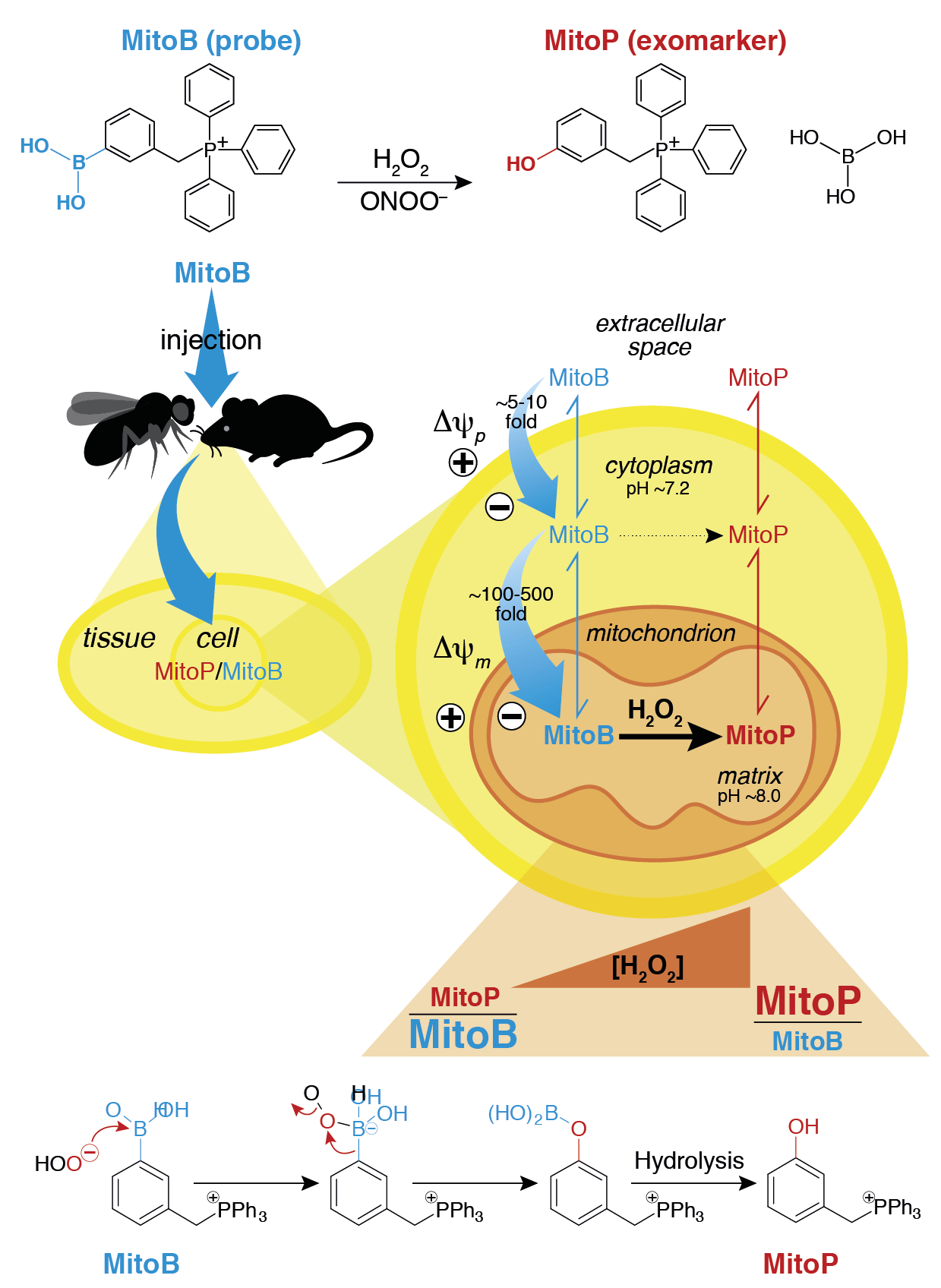
The ability to measure the concentrations of small damaging and signalling molecules such as reactive oxygen species (ROS) in vivo is essential to understanding their biological roles. While a range of methods can be applied to in vitro systems, measuring the levels and relative changes in reactive species in vivo is challenging. One approach towards achieving this goal is the use of exomarkers [1]. In this, exogenous probe compounds are administered to the intact organism and are then transformed by the reactive molecules in vivo to produce a diagnostic exomarker. The exomarker and the precursor probe can be analysed ex vivo to infer the identity and amounts of the reactive species present in vivo. This is akin to the measurement of biomarkers produced by the interaction of reactive species with endogenous biomolecules. We have developed mitochondria-targeted probes that generate exomarkers that can be analysed by mass spectrometry to assess levels of reactive species within mitochondria in vivo.
We have used one of these compounds, MitoB, to infer the levels of mitochondrial hydrogen peroxide within flies and mice [2]. This has been extended to superoxide (NeoD and MitoNeoD) [3], hydrogen sulphide (MitoA) [4] and glyoxals (MitoG and MitoGamide) [5].
This exomarker approach has been extended to other mitochondrial parameters such as the mitochondrial membrane potential. We have developed two mitochondria-targeted 'Click' reagents that both get accumulated ~1,000-fold by mitochondria in response to the membrane potential. There they react with each other ~1,000,000 faster than would occur extracellularly. Quantification of the product of this Click reaction by mass spectrometry allows a read out of mitochondrial membrane potential in vivo [6].
REFERENCES
- Logan A, Cochemé HM, Pun PBoon Li, Apostolova N, Smith RAJ, Larsen L, Larsen DS, James AM, Fearnley IM, Rogatti S, Prime TA, Finichiu PG, Dare A, Chouchani ET, Pell VR, Methner C, Quin C, McQuaker SJ, Krieg T, Hartley RC & Murphy MP (2014)
Using exomarkers to assess mitochondrial reactive species in vivo.
Biochim Biophys Acta 1840, 923-30 - Cochemé HM, Quin C, McQuaker SJ, Cabreiro F, Logan A, Prime TA, Abakumova I, Patel JV, Fearnley IM, James AM, Porteous CM, Smith RAJ, Saeed S, Carré JE, Singer M, Gems D, Hartley RC, Partridge L & Murphy MP (2011)
Measurement of H2O2 within living Drosophila during aging using a ratiometric mass spectrometry probe targeted to the mitochondrial matrix.
Cell Metab 13, 340-50 - Shchepinova MM, Cairns AG, Prime TA, Logan A, James AM, Hall AR, Vidoni S, Arndt S, Caldwell ST, Prag HA, Pell VR, Krieg T, Mulvey JF, Yadav P, Cobley JN, Bright TP, Senn HM, Anderson RF, Murphy MP & Hartley RC (2017)
MitoNeoD: A Mitochondria-Targeted Superoxide Probe.
Cell Chem Biol 24, 1285-1298 - Arndt S, Baeza-Garza CD, Logan A, Rosa T, Wedmann R, Prime TA, Martin JL, Saeb-Parsy K, Krieg T, Filipovic MR, Hartley RC & Murphy MP (2017)
Assessment of H2S in vivo using the newly developed mitochondria-targeted mass spectrometry probe MitoA.
J Biol Chem 292, 7761-7773 - Pun PBoon Li, Logan A, Darley-Usmar V, Chacko B, Johnson MS, Huang GW, Rogatti S, Prime TA, Methner C, Krieg T, Fearnley IM, Larsen L, Larsen DS, Menger KE, Collins Y, James AM, Kumar GDKishore, Hartley RC, Smith RAJ & Murphy MP (2014)
A mitochondria-targeted mass spectrometry probe to detect glyoxals: implications for diabetes.
Free Radic Biol Med 67, 437-50 - Logan A, Pell VR, Shaffer KJ, Evans C, Stanley NJ, Robb EL, Prime TA, Chouchani ET, Cochemé HM, Fearnley IM, Vidoni S, James AM, Porteous CM, Partridge L, Krieg T, Smith RAJ & Murphy MP (2015)
Assessing the Mitochondrial Membrane Potential in Cells and In Vivo using Targeted Click Chemistry and Mass Spectrometry.
Cell Metab 23, 379-385